Steps for sucessful atrial pacing
Atrial Pacemaker
While dual chamber pacing is preferred for valid indications, AAIR pacing remains a not so infrequently used option.
Introduction
RV pacing provides life sustaining rhythm support and is the most critical lead of a pacing system. However more and more patients are getting paced for non-life threatening yet highly symptomatic sinus node dysfunction. Therefore functional pacing and maintenance of physiologic cardiac activation is a prime concern when pacing for sinus dysfunction. To enable such, atrial pacing has evolved to be an integral component of pacing for sinus node dysfunction.
As long-term RV pacing has been proven to be deleterious, it became unwise to pace the RV when the problem is in the sinus node. Therefore initial attempts to avoid RV pacing was to pace only the atrium provided the AV nodal function was acceptable. This was the AAI mode. [ Atrial demand pacing]. Subsequent development of advanced sensor technology enabled physiologic rate adaptation mimicking natural sinus node activity and AAI became AAIR [ Rate adaptive Atrial Demand Pacing]. Atrial only pacing was possible only if the AV node was normal – otherwise had to pace the ventricle. On the west side of the Atlantic, the skepticism for atrial only pacing was high and this accelerated the development of dual chamber pacing systems which would pace both chambers based on the underlying pathology and rhythm.
Dual chamber systems introduced an important benefit – maintenance of AV synchrony. This enabled AV synchronized pacing for even AV nodal disease (e.g. complete heart block) in addition to sinus node dysfunction. The biggest perceived advantage of dual chamber pacing in sinus disease was that if the patient developed AV nodal disease, there would be synchronized pacing for ventricles too. Dual chamber pacing became very popular in North America and was the mode of choice for pacing for a wide variety of brady-arrhythmias except permanent slow AF. However the Europeans were skeptical of dual chamber pacing and until DANPACE shed light to the matter, atrial only pacing remained quite popular in Europe. DANPACE provided evidence base for justification of dual chamber pacing for sinus node dysfunction – for clinico-pathological reasons and economic reasons
Update 2022. The newly released 2021 ESC pacing guideline does away with AAI pacing in preference to DDD. However the 2018 AHA guideline still has a Class 1 recommendation for AAI pacing in patients with pure sinus node dysfunction.
Regardless of the above, dual chamber systems are still very expensive compared to single chamber systems (and there is a unresolved issue of risk of heart failure with pure sinus node dysfunction). Therefore in cost-constrained developing country environments such as ours, we still attempt AAIR pacing if feasible. Usually these patients are young patients with only sinus node dysfunction and have had a non – invasive workup to assess AV nodal function before pacing AAI. An intra-operative testing of AV nodal function determines the final mode of pacing (our protocol is given in the section on Atrial Lead Implantation)
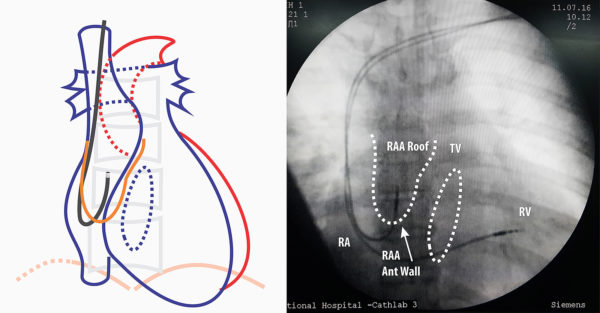
With regard to the site of pacing, right atrial appendage (RAA) is the location of choice – mainly because its an easily accessible and stable place. We avoid the lateral atrial wall because of phrenic nerve capture (and rare “it has happened” risk of bleeding), The inter-atrial septum is only very rarely used for pacing because almost always, a good site in the RAA can be found – even is situations where the RA appendage might have been partially amputated by the cardiac surgeons
Key Documents
Recommended Reading
- Kenneth A. Ellenbogen – Clinical Cardiac Pacing, Defibrillation, and Resynchronization Therapy 4e.
- Chapter 10 : Clinical Trials of Atrial and Ventricular Pacing Modes by Carsten W. Israel
- Fred M. Kusumoto – Cardiac Pacing for the Clinician 2e. –
- Chapter 9 : Sinus Node Dysfunction by Irene H. Stevenson, Paul B. Sparks, and Jonathan M. Kalman
- Chapter 10 : Acquired Atrioventricular Block by S. Serge Barold and Bengt Herweg
- Chapter 9 : Sinus Node Dysfunction by Irene H. Stevenson, Paul B. Sparks, and Jonathan M. Kalman
Implantation of Atrial Lead
The steps are
- Have vascular access : Either the only sheath (for AAIR) or second sheath for a dual system
- If dual – ventricle lead in place and finalized
- Insert atrial lead and park in right atrium (as described for the ventricular lead)
- Change the straight stylet with a appropriate J curved stylet
- Negotiate the distal curved end of the lead to the Right Atrial Appendage under fluroscopic guidance
- Check parameters and deploy active fix screw
- Exchange stylet to a straight one and anchor the lead at the pocket after removing the sheath.
Firstly, the lead has to be parked in the RA. If the system is going to be only AAIR then, the existing sheath can be used to insert the lead as described here for ventricular pacing. If it is a dual chamber system, an additional sheath has be in place as described here via an additional guide-wire as described here.
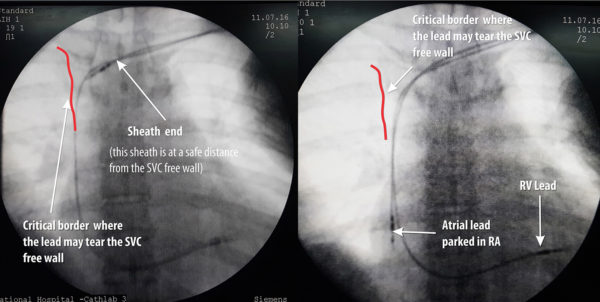
One potential place where brisk forcible insertion might cause problems is the SVC free wall: If the sheath is too much in, the tip will lie against the free wall of the SVC and the lead coming out of it will impinge on the wall creating a resistance. Therefore it is imperative that fluroscopic screening is done at the slightest resistance without pushing the lead blindly. If impinging, the whole sheath should be slightly withdrawn and the lead marched forward with about inch of stylet pulled out to make it floppy and fall down
Figure : Precautionary measure when inserting lead (AP view) – It important to check the sheath tip position and insert the lead. If there is resistance, fluroscopy is essential before advancing the lead (see text)
Figure : Lead Guided to right atrial appendage AP View- Next step is to remove the straight stylet and insert the J stylet. J – stylet has to be inserted slowly as when the stylet reaches end, the lead assumes a J- shape and this can impinge on right atrial wall. Once the J is visible, the whole lead is slowly pulled up as to make it “hook” into the roof of RA appendage. If there is sinus node activity, the distal end of the lead will be seen moving from side to side as in car windshield wiper “hence called the wiper movement of the atrial lead”
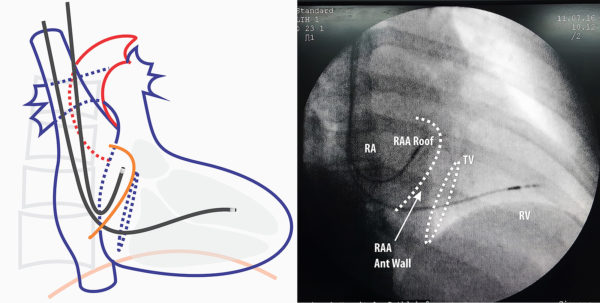
Figure : Lead Guided to right atrial appendage RAO view – If one is not sure about the position, RAO will confirm the anterior nature of the lead tip if it is correctly in the appendage.
Video : Positioning of atrial lead. The lead should be inserted along with same precautions for the RV lead. Once in RA, the straight stylet is exchanged for a J- Shaped stylet. Once the lead is in the appendage, it will have sideways wiper like movements. However these movements may be absent if the patient has no P waves. Once the lead is in palace, electrical parameters can be checked and the lead screw deployed
Note : The atrial leads are usually shorter than the ventricular leads. For a given manufacturer, all pacing leads are electrically identical but differ in lengths to facilitate ventricular versus atrial pacing. However they are interchangeable (e.g. A longer ventricular lead can be used for atrial pacing vice versa). Medtronic, St Jude Medical and Vitatron have 58 cm leads for the ventricles and 52 cm leads for the atria. Biotronik has 60 cm leads for the ventricle and 53 cm leads for the atria
Each lead is accompanied by a one or two pre-formed J shaped stylets – these vary in size and stiffness. Therefore if a lead refuses to move into the appendage, a stylet with a smaller curve r different stiffness can be be tried.
Video : Active fixation of atrial lead . Once parameters are confirmed, the active fixation screw is deployed. It is important to note that a tiny pull of 1 to 2 mm is applied to the lead (in contrast to the push when deploying the RV lead) midway of screw deployment – this pulls the lead tip against the atrial RAA roof to ensure a good fix. After deployment, the J stylet is withdrawn and it is imperative that this step be done very slowly. (see below)
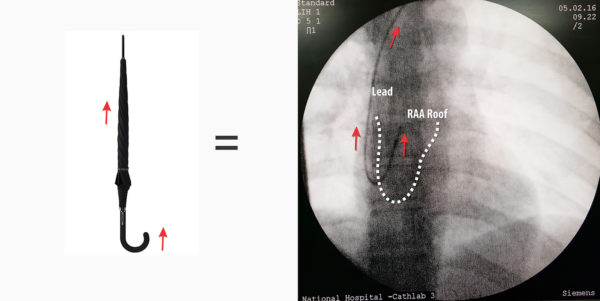
Video : Removal of the J-tip stylet . After screw deployment, the J tip stylus needs to be removed in a controlled fashion – this is to ensure that it does not get dislodged or worse – cause a bleeding. The lead body must be firmly held by one hand and the stylet removed slowly (See below) . If you don’t hold the lead firmly, pulling of the stylet will pull the entire lead as the J curve acts like an umbrella hook on a nostril! Slow removal helps the stylet to slide out instead of pulling the whole lead along with it. Furthermore, just before pulling the stylet I push the lead a bit to the atrium to give it more slack to facilitate the stylet coming out.
If one appreciates the shape of the lead which is given by the stylet) it can be easily figured out why it is dangerous to pull the stylet fast : It can lead to a lead out-migration through the appendage roof. If pulled fast, the J stylet tip provides force at the lead tip forcing it cranially against the appendage roof. Since the screw is already deployed, it gets further wedged in the muscle wall and if the site of anchoring has a thin wall, the deployed screw length is sufficient to reach out of the myocardium into the pericardium. The potential serious complication is bleeding to the pericardial space – but the usual symptom is central pricking sharp chest pain. Author has seen three such cases where symptoms rapidly resolved after re-positioning the atrial lead.
Figure : Cranially directed force from rapid withdrawal of stylet. Note the red arrow depicts a rapid forceful pull. If pulled slowly and gently, the stylet has time to unwind slide out of the lead lumen
Figure : Technique for withdrawing the J-Stylet from an atrial lead. One hand is used to hold the lead tightly in place and the other hand to pull out the stylet. Initial withdrawal has to be very slow. It should be done under fluroscopy because witnessing the wriggling movement of the lead while stylet comes out confirms good fixation of the lead.
Video: Final appearance of atrial pacemaker. Note the slack on the atrial lead to allow for any post implantation lead pulling.
Unexplained central pricking type chest pain after atrial pacing should prompt lead re-positioning. If the patient is on anticoagulants or anti-platelets, they should be temporarily withheld if not absolutely necessary – even in the absence of acute pericardial bleeding – as delayed bleeding may occur. Such patients warrant an echocardiograph in the ward.
So how to decide on AAI ?
If the pacing is for pure sinus node dysfunction, AAIR pacing can be considered (although dual chamber pacing is preferred). To pace in AAIR, one has to be absolutely sure that there is no current AV nodal disease. We assess this in two ways – Non invasively with Holter and Exercise testing and invasively during implantation of device. If holter and exercise ECG shows only evidence of sinus node dysfunction, we plan for an AAIR system and do intra-operative reconfirmation of AV nodal function. The intra-operative method is described below.
AV Nodal Wenckebach Point : AV node has decremental conducting properties i.e. at progressively faster sinus rates, the conduction time over the AV node increases (with manifest PR prolongation) and after a certain rate, it begins to block the conduction of certain P waves. This occurs in a pattern where the PR interval progressively lengthens and suddenly fails to conduct a P wave. Soon after the blocked P the subsequent conducted P wave has a shorter PR interval than the last conducted P wave thus implying that the AV node recovered during the non -conducted P wave. This is similar to type 1 heart block (Wenckebach type) and the heart rate at which the first dropped beat occurred is termed the Wenckbach point (WBP)
AV nodal conduction is highly dependent on autonomic function. Increased vagal tone decreases conduction velocity and sympathetic stimulation increases conduction velocity. Although Wenchbach phenomenon occurs at fast heart rates, the phenomenon is often seen in healthy young physically fit adults during sleep as their vagal tone is high during sleep. AV nodal conduction is also influenced by drugs such as some calcium channel blockers, digoxin, beta blockers etc.
In purely sick sinus disease, the AV nodal conduction is assumed to be normal. For pre-operative planning, we mainly do a holter and if the holter does not show evidence of AV nodal disease (baseline PR prolongation, Non conducted Ps in awake state) we consider AAI pacing. The issue with exercise testing is that due to sinus node disease, most patients are unable to mount a good sinus response to exercise – hence are unable to do an exercise ECG – but rarely a patient may have good chronotropic competence and if physically capable, doing an exercise ECG may provide useful information.
Note : The clinical context also has to be considered. If there is a significant burden of AT/AF which requires significant rate controlling medications (e.g. Betblockers, Verpamail, Diltiazem or digoxin) then AAI may not be a good option. If the primary strategy is rhythm control in PAF, then AAI may be a suitable option – provided that excessive nodal drugs are not used.
If AAI is anticipated, following steps are taken
Video : Intra-operative testing for AV node conduction – Atrial pacing rate is gradually increased and AV conduction is observed and in this example a regular missed conducted beats (Wenckbach phenomenon ) is seen at atrial rates faster than 130.